Container Closure Integrity Testing (CCIT)
Container Closure Integrity Testing (CCIT) is a critical evaluation of container closure systems’ ability to maintain a sterile barrier against contamination, ensuring the safety and stability of pharmaceutical, biological and vaccine products.

It’s vital to perform CCIT to prevent issues with pharmaceutical products. Container closure integrity defects can alter the active pharmaceutical ingredient (API) due to loss of vacuum, oxidation, hydrolysis, or bacterial contamination. This compromises efficacy, causing unexpected side-effects and harming patient health. Sepha offers Vacuum Decay, Pressure Decay and Vacuum Deflection solutions.
What is a Container Closure Integrity Test?
A Container Closure Integrity Test assesses the integrity of container closures to prevent content leakage and the ingress of moisture, air, or contaminants.
It involves evaluating the integrity of container closure systems to maintain a sterile barrier against potential contaminants. This testing is essential because even minuscule defects, such as pinholes measuring less than 1000th of a millimeter, can compromise the sterility and stability of pharmaceutical products. Regulatory bodies like the US Food and Drug Administration (FDA) recognize these defects as serious threats to product safety.

What are the types of container closure integrity tests?
Various CCIT methods are available, some with ASTM standards, which are widely recognized for regulatory compliance.
Probablisitic methods including Blue Dye Methylene, bubble emission (ASTM D3078-02 (2013), microbial challenge by immersion and tracer gas detection, sniffer mode (ASTM F2391-05 (2016) are the more traditional methods that are still widely used.
However, in recent years, deterministic methods have gained favour – regardless of whether they have an ASTM standard associated with them – due to their accuracy and reliability, compared to probabilistic methods. Deterministic methods including vacuum decay (ASTM F2338-09), pressure decay, Headspace Gas Analysis, HVLD, Vacuum Deflection by laser measurement (ASTM F3169-16) and tracer gas analysis (vacuum mode: ASTM F2391-05 (2016)) offer non-destructive testing options and objective measurements of leakage.

What types of containers can be tested with CCIT methods?
CCIT methods are versatile and can be applied to a wide range of pharmaceutical containers, ensuring that the integrity of each container is thoroughly evaluated.
This includes rigid containers like glass and plastic vials, ampoules, pre-filled syringes, cartridges, blow fill seal strips, and bottles, but also solid oral dosage packaging including blister packs. These methods are crucial, particularly for injectable, parenteral products, where maintaining sterility is of utmost importance.
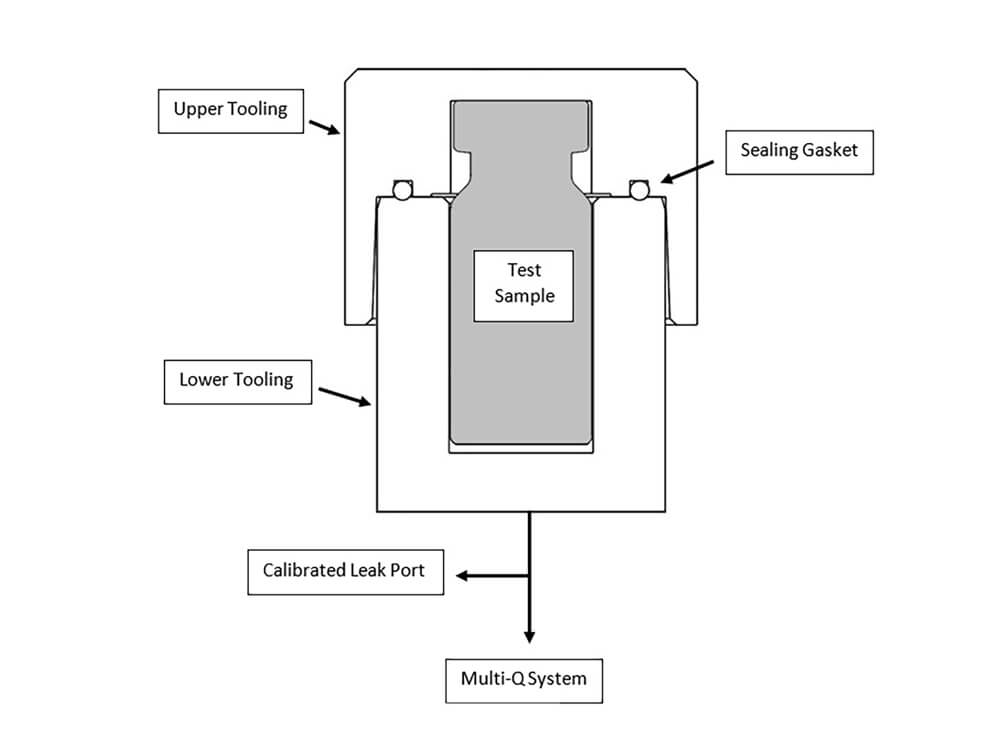
How do you perform a container closure integrity test?
The testing process varies based on the method used but generally aims to identify any breach in the sterile barrier.
Sepha offers a range of Container Closure Integrity Tests, including Vacuum Decay and Pressure Decay testing for parenteral containers, and Vacuum Deflection for pharmaceutical blister packs to ensure package integrity, ultimately safeguarding patient health and brand reputation.
- To perform a vacuum decay test, a container is placed inside a sealed chamber which is evacuated to a predetermined vacuum level. A sensor then monitors the vacuum over a set time. If there’s a leak, the pressure will rise, indicating a gross, medium, or micron leak based on predetermined vacuum levels and decay limits.
- To conduct a pressure decay test, a container is placed in a test chamber that is pressurised to a target level. The chamber is then allowed to stabilize, and the decay or loss of pressure is measured over a set period of time. Any change in pressure indicates air is leaking into the container being tested, indicating the presence of a leak.
- To conduct a vacuum deflection test, a blister pack is placed inside a vacuum test chamber and scanned by a laser to establish initial data points. Then, a vacuum is applied, and the pack’s surface is scanned again. The deflection, or height difference in the foil of a cavity under vacuum, is measured. If there’s a large hole in the cavity, there won’t be significant deflection as pressure equalizes. For cavities with no holes, the foil moves, causing deflection. Small holes cause initial expansion under vacuum. A third scan is needed to identify these holes. Increased collapse during reduced vacuum signals the presence of a small hole, where air escapes gradually, equalizing the pressure inside the pocket with the applied vacuum.
Our Products

Multi-Q Vacuum Decay

Multi-Q Pressure Decay

Multi-Q HD
VisionScan 3D

Book free Multi-Q demo
To see a demo of the Multi-Q join us in Hall 3.0, stand A48. Don’t miss out, get in touch and get your free ticket below.
